 
if you know the strength you can work back to the ethanol density. AlcoDens can also be used in reverse mode, i.e. See Water - Enthalpy and Entropy for tabulated values in different units. If the ethanol density is known in any of these units AlcoDens can be used to determine the strength of the ethanol-water mixture over the range of temperatures from -20☌ to 100☌ (-4☏ to 212☏). Specific entropy of water (liquid and vapor) as function of temperature (☌ and ☏): See Water - Thermal Diffusivity for variations at constant pressures. 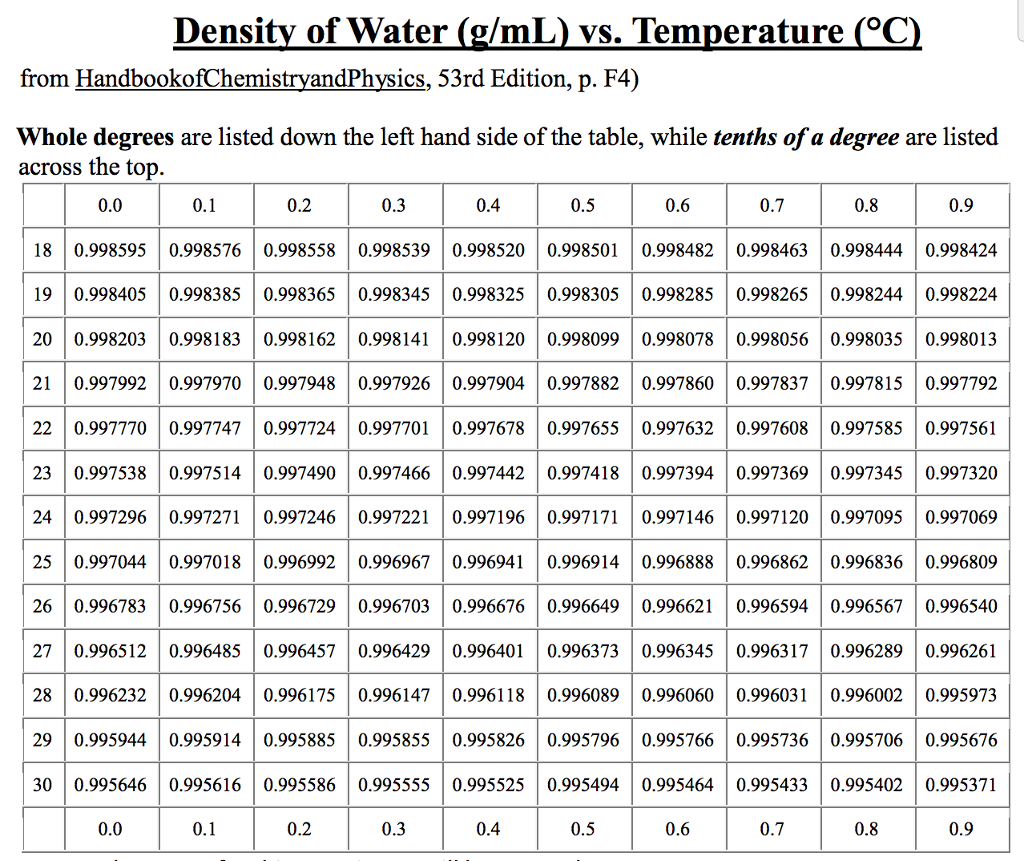
Thermal diffusivity of water (liquid and vapor) as function of temperature (☌ and ☏): See Water - Prandtl Number for variations at constant pressures. Prandtl number of water (liquid and vapor) as function of temperature (☌ and ☏): See Water - Thermal Conductivity for variations at constant pressures. The density of water (H 2 O) changes depending on temperature and pressure - as with other substances. Thermal conductivity of water (liquid and vapor) as function of temperature (☌ and ☏): See Water - Specific heat (heat capacity) for calculator of heat capacity at given temperatures. Specific heat (heat capacity) of water (liquid and vapor) as function of temperature (☌ and ☏): See Water - Density, Specific Weight and Thermal Expantion Coefficient for calculator and variations at constant pressures. See Water - Dynamic and Kinematic Viscosity for calculator and variations at constant pressures.ĭensity of water (liquid and vapor) as function of temperature (☌ and ☏): See Water - Saturation pressure for calculator of equilibrium pressure at varying temperature.ĭynamic viscosity of water (liquid and vapor) as function of temperature (☌ and ☏): Vapor pressure of water as function of temperature (☌ and ☏): See also these and other properties of Water at varying temperature and pressure : Boiling points at high pressure, Boiling points at vacuum pressure, Density and specific weight, Dynamic and kinematic viscosity, Enthalpy and entropy, Heat of vaporization, Ionization Constant, pK w, of normal and heavy water, Melting points at high pressure, Prandtl number, Properties at Gas-Liquid Equilibrium Conditions, Saturation pressure, Specific gravity, Specific heat (heat capacity), Specific volume, Thermal conductivity, Thermal diffusivity and Vapour pressure at gas-liquid equilibrium. See Water and Heavy Water for thermodynamic properties at standard condtions. Water is a pure liquid, and youll need to use the density to first convert volume to mass. HINT: Since water is not a solution, molarity does not apply. Tabulated values are given below the figures, in SI and Imperial units. Given: the density of water at room temperature is 0.998 g / mL. The figures show liquid and gaseous states at equilibrium as a function of temperature, starting at the triple point and ending at the critical temperature. The figures and tables below summarize the thermophysical properties of water and steam at equilibrium. At this equilibrium condition the vapor pressure is the saturation pressure. At higher pressures water would condense. The vapor pressure of water is the pressure at which water vapor is in thermodynamic equilibrium with its condensed state. If the space is confined the partial pressure exerted by the molecules increases until the rate at which molecules reenter the liquid is equal to the rate at which they leave. I'll go with $$20^$$.Water tends to evaporate or vaporize by projecting molecules into the space above its surface. The first thing to do here is to pick a temperature that can be considered room temperature.
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
